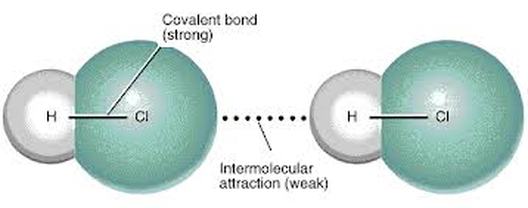
The uneven sharing of electrons causes a polar-covalent bond, a bond where one end is slightly negative and the other slightly positive. Dipole-dipole forces result from the attraction of the positive region of one to the negative region of a second molecule.
Since the dipoles of polar-covalent bonded atoms are permanent then dipole-dipole forces are stronger than dispersion forces.
Examples: Acetone in acetone, triethyl amine in acetone, ...
Since the dipoles of polar-covalent bonded atoms are permanent then dipole-dipole forces are stronger than dispersion forces.
Examples: Acetone in acetone, triethyl amine in acetone, ...