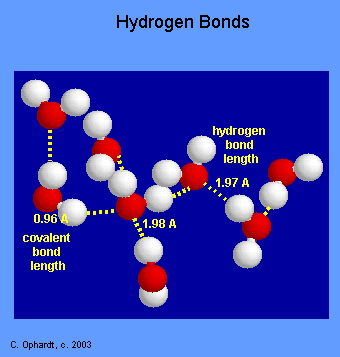
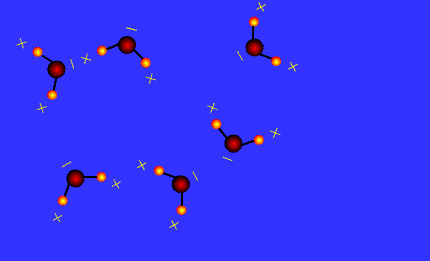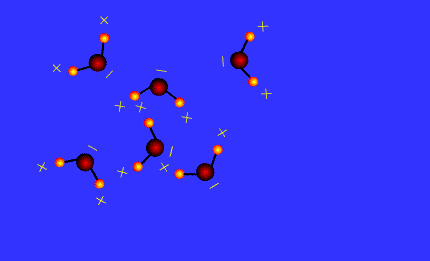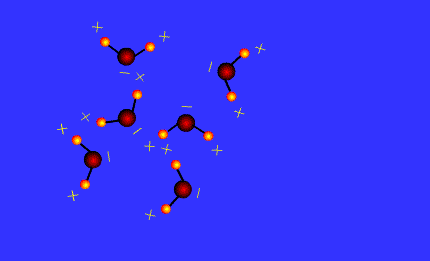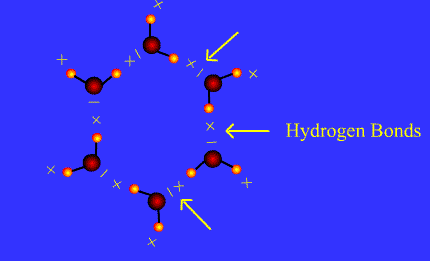
A hydrogen bond is a dipole-dipole attraction (special type). It takes place between molecules containing a hydrogen atom bonded to an atome with higher electronegativity with atleast one electron pair. Hydrogen must bond with either Fluorine, Oxygen, or Nitrogen atom inorder to form a hydrogen bond because these atoms are electronegative enough to cause a large partial positive charge on the hydrogen.
Hydrogen bonds are the strongest intermolecular force. Some examples are: water, acetic acid, acetone in water
Hydrogen bonds are the strongest intermolecular force. Some examples are: water, acetic acid, acetone in water