|
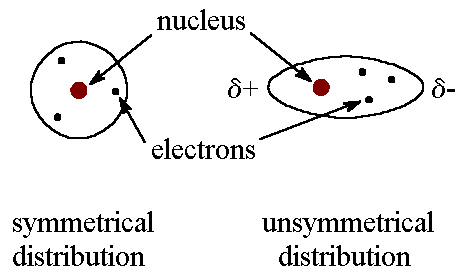
Dispersion forces are weak forces that result from temporary shifts in the density of electrons in the electron clouds and only between two non-polar molecules.
How? When two non-polar molecules are in close contact (especially when they collide) the electron cloud of one molecule repels the cloud of the other. However, the electron density around each nucleus is greater in one region of each cloud causing each molecule to form a temporary dipole meaning that a part of the molecule has a slight positive charge and the other part a slight negative. Examples : liquid methane gas, ... |
|
A weak dispersion force exists between oppositely charged regions of the dipole when temporary dipoles become close to each other.
Dispersion forces are the weakest intermolecular force due to the temporary nature of the dipoles. But on the other hand, is its the dominant force of attraction between identical non-polar molecules. |